We are pleased to announce that the first peer reviewed journal paper by researchers of the SOCRATES project has been accepted for publication! Researchers of the SOCRATES project from Outotec Oy and Aalto University are developing original process for taking high-purity gold product out of complex hydrometallurgical solution. The proof-of-concept paper has now been published in an Open Access in the Journal of Cleaner Production.
Selective recovery of gold with novel electrochemical method
Paper résumé
For more than a century, cyanide leaching has been the most common process for extraction of gold from ores and concentrates. At the same time, cyanidation has been blamed throughout the years for its hazardous impact on human health and environment. These concerns have given a rise for cleaner, cyanide-free alternatives, e.g. chloride leaching. However, the state-of-art processes for gold recovery from chloride solutions are underdeveloped and facing issues of high reagent consumption and poor selectivity.
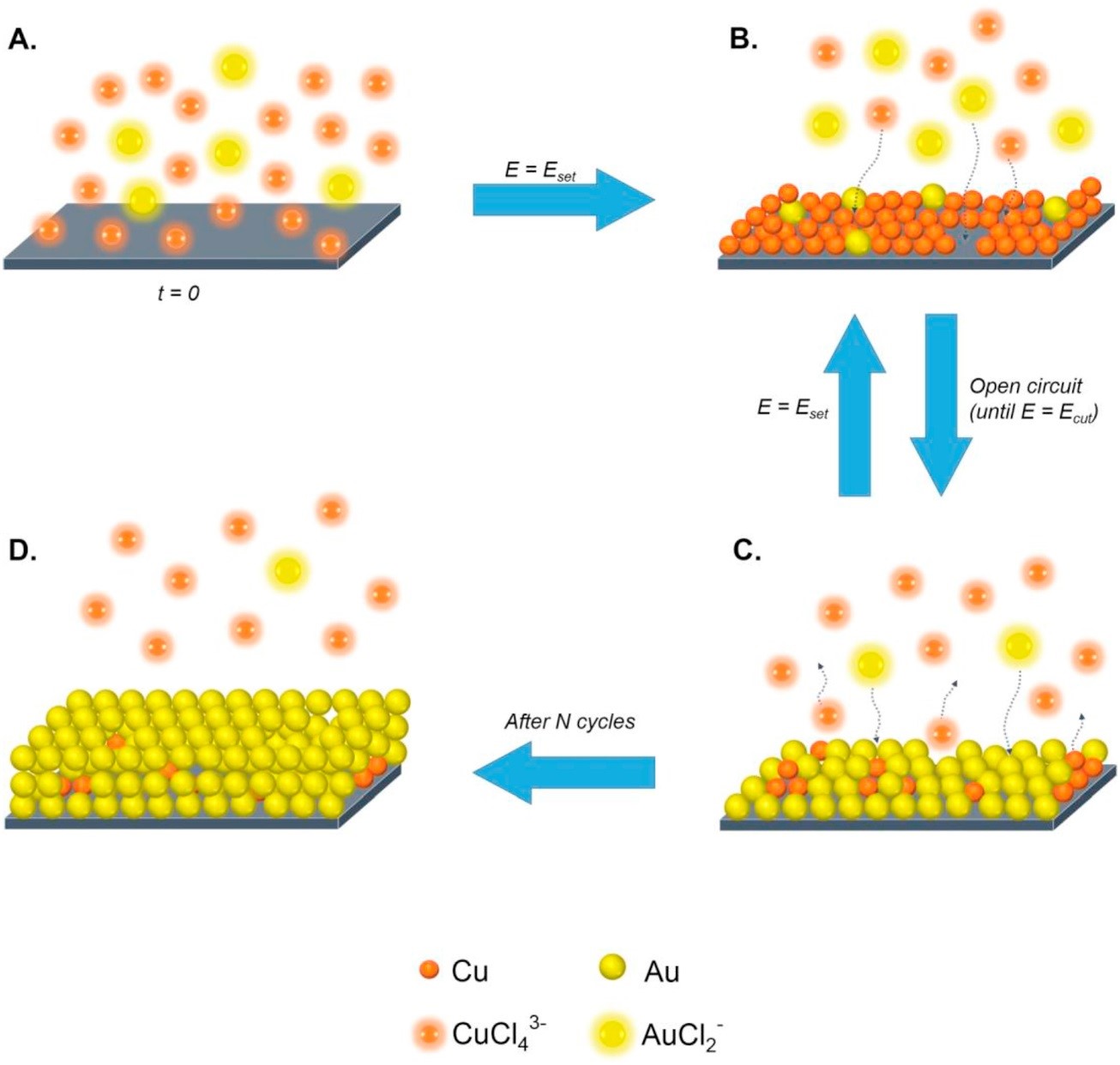
In this paper, the authors describe an innovative concept for recovery of minor concentrations of gold from hydrometallurgical solutions by repetitive Electrochemical Deposition-Redox Replacement (EDRR) cycles. In contrast to conventional adsorption technologies or solvent extraction, the proposed electrochemical method does not require addition of any chemicals in the process to achieve remarkably selective gold recovery. The final gold product was analyzed by SEM, XPS and ultimately by dissolution in aqua regia. Results indicate that the Au content in the metal deposit after 250 EDRR cycles (ca. 24 hours) exceeded 75% and the Au:Cu ratio has increased by a factor of 1000, from 1:340 in the solution up to 3.3:1 in the final product.
All in all, the obtained results prove that the EDRR method can be productively used for the recovery of trace amounts of gold from cupric chloride solutions used for cyanide-free gold leaching in environmentally friendly and resource-efficient manner. Utilizing EDRR could reduce the need for resources and the costs related to entire gold recovery process. However, the detailed investigation of energy efficiency and energy consumption of this particular process is the subject of future studies.
Full reference paper
Ivan Korolev, Pelin Altinkaya, Petteri Halli, Pyry-Mikko Hannula, Kirsi Yliniemi, Mari Lundström, Electrochemical recovery of minor concentrations of gold from cyanide-free cupric chloride leaching solutions, Journal of Cleaner Production 186 (2018) 840–850, DOI: 10.1016/j.jclepro.2018.03.177
Acknowledgements
The research leading to these results received funding from the EU Framework Programme for Research and Innovation Horizon 2020 under Grant Agreement No 721385 (EU MSCA-ETN SOCRATES).